 See also laser.įacts You Should Know: The Periodic Table Quiz This bundle is known as a photon, and this emission of photons with a change of energy levels is the process by which atoms emit light. For example, if an electron jumps from a higher to a lower energy level, the lost energy will have to go somewhere and in fact will be emitted by the atom in a bundle of electromagnetic radiation. This process is called a quantum leap or quantum jump, and it has no analog in the macroscopic world.īecause different orbits have different energies, whenever a quantum leap occurs, the energy possessed by the electron will be different after the jump. An electron disappears from the orbit in which it is located and reappears in its new location without ever appearing any place in between. As with many processes in the quantum world, this process is impossible to visualize. The laws of quantum mechanics describe the process by which electrons can move from one allowed orbit, or energy level, to another. The orbits are analogous to a set of stairs in which the gravitational potential energy is different for each step and in which a ball can be found on any step but never in between. In the Bohr atom electrons can be found only in allowed orbits, and these allowed orbits are at different energies. This property, first explained by Danish physicist Niels Bohr in 1913, is another result of quantum mechanics-specifically, the requirement that the angular momentum of an electron in orbit, like everything else in the quantum world, come in discrete bundles called quanta. 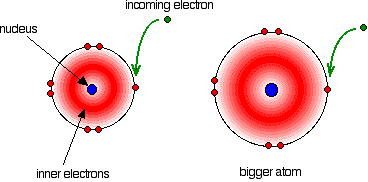
Unlike planets orbiting the Sun, electrons cannot be at any arbitrary distance from the nucleus they can exist only in certain specific locations called allowed orbits.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
